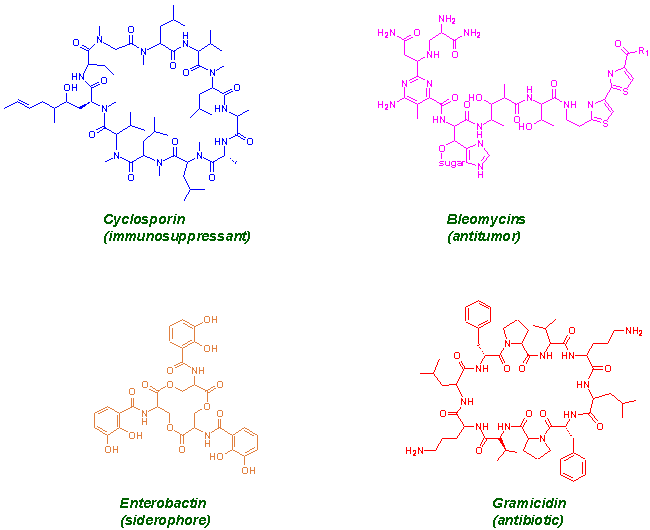
Nonribosomal peptides belong to a family of complex natural products built from simple amino acid monomers. They are synthesized in many bacteria and fungi by large multifunctional proteins called nonribosomal peptide synthetases (NRPS). A unique feature of NRPS system is the ability to synthesize peptides containing proteinogenic as well as non-proteinogenic amino acids.In many cases these enzymes work in conjunction with polyketides synthases (PKS) giving hybrid products. The product of these multifunctional enzymes are very important pharmaceutical compounds like cyclosporin, gramicidin etc.

NRPSs are organized into coordinated groups of active sites termed modules, in which each module is required for catalyzing one single cycle of product length elongation and modification of that functional group. The number and order of module and the type of domains present within a module on each NRPS determines the structural variation of the resulting peptide product (Fig 1) by dictating the number, order, choice of the amino acid to be incorporated and the modification associated with a particular type of elongation.
The minimum set of domains required for an elongation cycle consist of a module with Adenylation(A), Thiolation(T) or Peptidyl Carrier Protein (PCP), and Condensation(C) domain.
CATALYTIC DOMAINS:
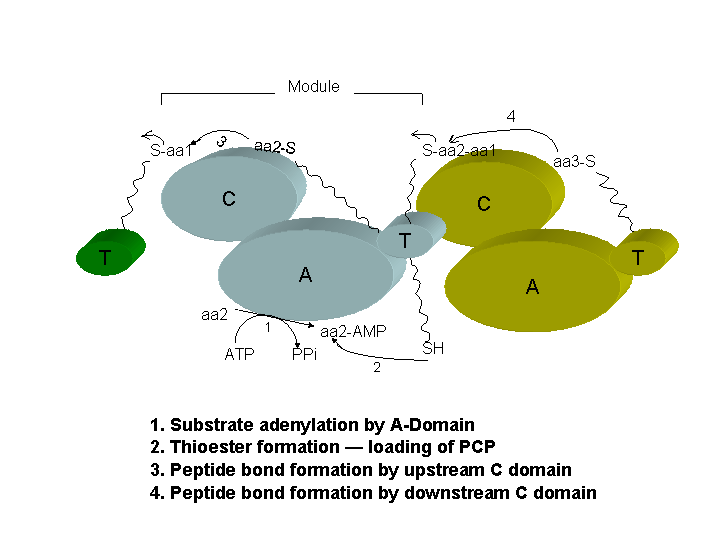
The Adenylation domain is responsible for substrate
selection and its covalent fixation on the phospho-pantethein arm of T
domain as thioester, through AMP-derivative intermediate.
The C domain catalyzes the formation of peptide bond between an aminoacyl- or peptidyl-S-PCP from the upstream module and the aminoacyl moeity attached to the PCP in the cooresponding downstream module. The result is peptide elongation by one residuefixed to the PCP domain in the downstream module. Optional modifying domain could be present for substrate epimerization, N-methylation and heterocyclization. The modules could remain on a single or multiple polypeptide chains.
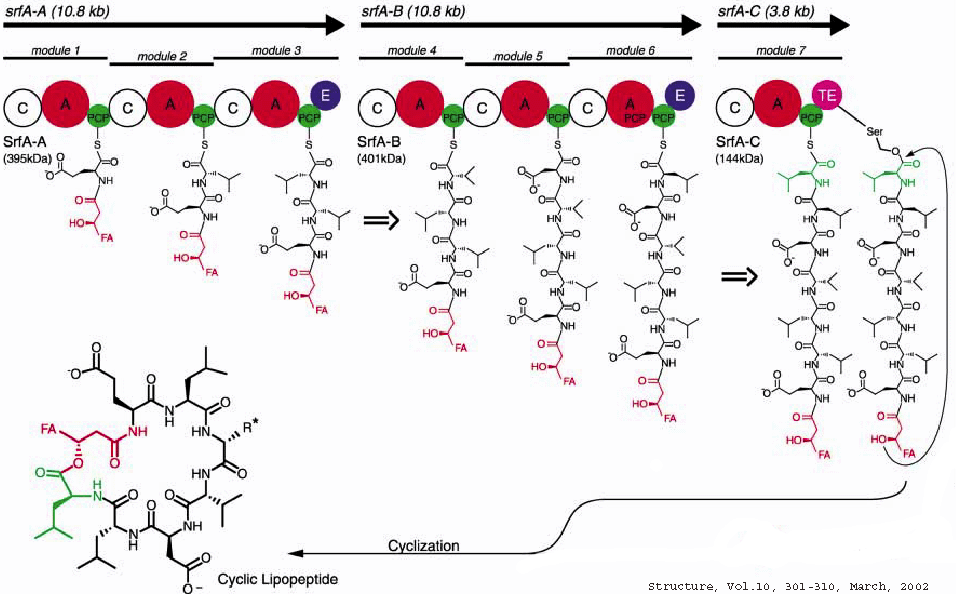
In most cases, there is an extreme C-terminal Thioesterase(TE)
domain in the last module responsible for the release/cyclization of the
final product.